|
As chief hydrologist in France during
the first half of the last century, Professor Vincent had the unique
opportunity to gather statistics showing disease tendencies and
mortality rates in populations across Europe.
Professor Vincent correlated the electrical values of
pH (acid/alkaline balance), rH2 (oxidation--reduction potential)
and R (resistivity - level of electrically conductive ions) to the body fluids of blood, urine and saliva.
He cross-correlated this information
to the water quality those same populations were consuming. Professor Vincent found populations with
alkaline and
oxidized tap water tended to have greater incidence of cancers
and thrombosis.
"The amount and type of water we drink is very important to our ability to maintain health.
Like food, water can be wholesome or can contribute to disease. The properties of the water we drink are as important as the nutritional content of the food we eat."
His research revealed a set of optimal values at the
intersection of pH, rH2 and resistivity
that was determined to be the most biocompatible for the human organism.
The best water for human consumption meets the Vincent (BEV) standards for purity and biocompatibility.
pH is a measure of acidity or alkalinity, i.e. the hydrogen ion concentration.
Assuming the rH2 and r values are within accepted values, the pH of bio-compatible water can range from 4.0 to 6.9.
This range is allowed because in extremely pure water it is very easy to effect the pH value—there are no other dissolved substances to buffer the solution.
Regrettably, many municipal systems add lime to raise the pH between 7.3 and 8.0 in order to protect pipes from corrosion.
(In fact, many of the toxic compounds found in drinking water come from the distribution system itself.
Some, like chlorine and fluoride, are intentionally added; others are acquired as water flows through aging and leaking underground pipes -
picking up traces of any number of metals including copper, nickel, chromium, even lead.)
rH2 (a calculated value that provides an indication of
true oxidation/reduction potential)
and tells you the vitality or energy potential of the water. rH2 also has a scale (similar to pH) that measures from 0 to 42,
with 28 representing the mid-point. Values above 28 are considered oxidizing, below 28 reducing.
The best water for human consumption has an rH2 slightly on the reducing side of the scale (20 - 24).
Unfortunately, the majority of chlorinated tap water supplies have an rH2 value higher than 31.
Resistivity (the opposite of conductance) is used to measure the amount of dissolved inorganic solids, or TDS.
The resistivity value should be as high as possible, indicating an exceptionally low concentration of dissolved contaminates.
The higher the resistivity value, the cleaner your water and the better it is for you.
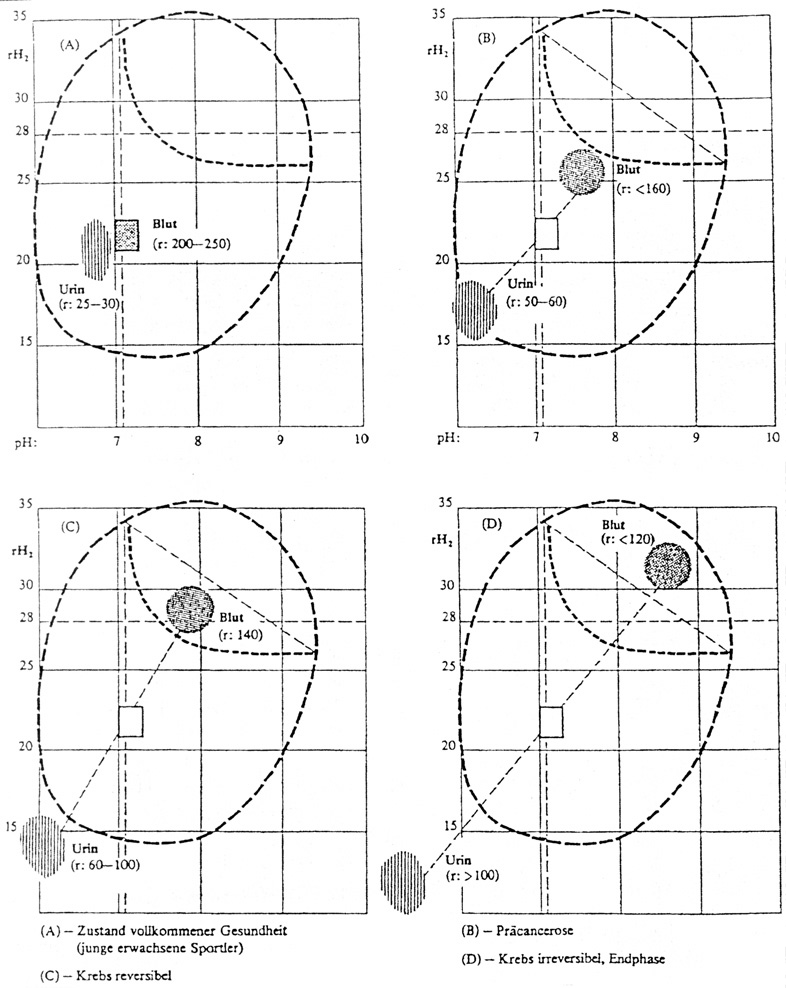
The following charts show blood and
urine values in the following groups.
(A) healthy, (B) precancerous, (C) reversible cancer, (D)
irreversible cancer
|
